PharmaJet Tropis ID: A proven solution for intradermal (ID) delivery – overcoming the challenges made evident by the Monkeypox (mpox) vaccine shortage.
Last summer, when monkeypox (mpox, in 2024) was declared a national1 and global health emergency, a shortage of the mpox vaccine was announced2. In August 2022, the US Food and Drug Administration (USFDA) authorized3 reduced dose (0.1 mL) intradermal (ID) delivery of mpox vaccine to address the supply gaps and extend limited stocks. The EU followed with their own recommendations on intradermal delivery. 4
What does “reduced dose” mean?
Reduced dose immunization, often referred to as “dose sparing” or “fractional dosing” is not new. Responding to the USFDA announcement, Dr. Bruce Weniger, an expert on the ID technique and retired CDC scientist confirmed, “The ID approach has been used to stretch vaccine supply for other diseases.”5 As an example, fractional dose has been successfully used for mass polio campaigns for years. A systematic review and meta-analysis published by The Lancet6 strengthened evidence that “20% dose delivery is a safe, immunogenic, and effective alternative to full dose inactivated polio vaccine (IPV). Immunization programs can achieve high levels of immunity against all poliovirus types by substituting fractional IPV (fIPV) for full dose IPV.”
In addition to polio vaccine, reduced dose regimens for rabies vaccination have been recommended by the World Health Organization (WHO) since 1992 and have been widely used in Southeast Asia (India, Thailand, etc.). In fact, over the past decades, numerous studies indicate that, for several vaccines, including hepatitis B, smallpox, and influenza, reduced dose immunization delivered intradermally exhibits similar, or even enhanced, immunogenicity, as compared to full dose intramuscular (IM) or subcutaneous (SC) immunization7. Considering fragile supply chains and uncertainty whether the global supplies of key vaccines can support rapid scaling, reduced dosing is an important strategy with precedence. Choosing reduced dose instead of full dose can stretch supplies and lower the cost of vaccination.
In each of these cases, polio, and rabies, reduced dosing is accomplished with ID delivery. The efficacy of ID delivery is due to the high density of several types of immune cells such as dendritic cells (DCs), T lymphocytes, natural killer cells, macrophages, antigen presenting cells (APCs), and mast cells in the epithelium. The Langerhans cells in the skin, responsible for triggering the inflammation cascade, comprise 2-4% of epithelial cells. Langerhans cells are a specific DC subset that migrate into the lymph node following antigen capture and aid in the initiation of an adaptive immune response. Due to abundant APCs in the dermis, ID delivery of reduced doses (most often 20% or 30% of the standard amount of antigen) can induce immune responses equivalent to standard doses delivered via IM or SC routes.
How do we achieve scale using intradermal delivery?
ID delivery with needles is technically difficult, slow, painful, and inconsistent leading William Schaffner, an infectious disease expert at the Vanderbilt University Medical Center to comment: “As it happens, performing intradermal inoculation is the most difficult way to inoculate anyone. It’s less an inoculation than it is, in my opinion, an art form.”8 The “Mantoux” technique involves guiding a needle into the thin ID space, and if the health care worker goes too deep and inserts the dose into fat, the patient might not have the correct immune response. Conversely if the needle is not inserted far enough, some of the vaccine could leak back out and the patient does not receive the correct dose. “The average nurse and doctor would need to receive special training before performing the procedure,” according to John Beigel, a microbiologist and infectious disease specialist at the National Institute of Allergy and Infectious Diseases (NIAID).8 Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota commented, “This is a well-recognized issue where intradermal injections can be tough to do, without adequate training.” More specific concerns have also been raised with ID delivery using Mantoux: “Our team is getting three to four [doses] currently,” said Jeffrey Duchin, health officer for Seattle and King County Health Department, who added that that rate is “problematic because the actual number of doses we are getting is lower than the official allocation which assumes five doses per vial.”9 As a result of these challenges, ID delivery with needle and syringe has seen very little adoption during the current mpox crisis.
Fortunately, there is an alternative for ID delivery. The PharmaJet Tropis® ID Needle-free Injection System (NFIS) has been demonstrated at scale for global polio immunizations, using reduced dose ID delivery. It is WHO-prequalified, has registrations in more than a dozen countries, and is manufactured in quantities ready to respond to the next pandemic.
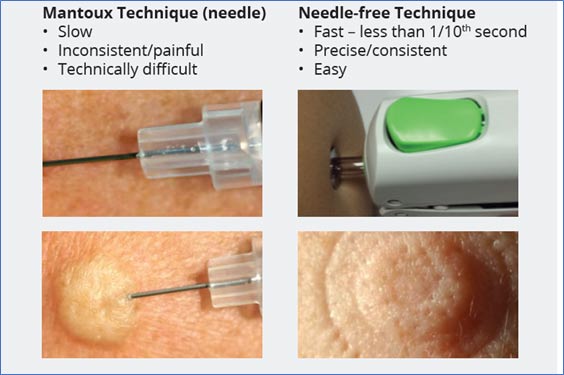
Comparing Needles to Needle-free ID Delivery
Needle-free is a better solution and is commercially available.
The PharmaJet Tropis NFIS was developed with support from the WHO, PATH, CDC, and Bill & Melinda Gates Foundation, as an alternative to needle and syringe (N/S). Tropis is an innovative, hand-held device that uses spring power to deliver a pressurized liquid stream without needles. When used in a WHO polio immunization campaign, vaccinators and caregivers who previously used N/S expressed a strong preference for the PharmaJet Tropis.10 In particular, they cited ease and speed of use and the added benefit of reduced discomfort and crying in the children receiving vaccinations. The PharmaJet NFIS has significantly faster preparation and administration time compared to the traditional ID technique. It requires minimal training, and minimizes vaccine wastage, making it an excellent tool for mass vaccination campaigns and routine use.
PharmaJet’s NFIS technology is commercially available and currently being used in over 80 research studies, 29 of which are clinical trials. The research applications of PharmaJet NFIS cover a number of disease areas and vaccine platforms including nucleic acids, inactivated, subunit, live attenuated, vial vector and bacteria.
Our partners have seen improved effectiveness, consistency of delivery, less wastage, and ease of training and implementation with Tropis ID. The chart below highlights the results from campaigns in The Gambia, Cuba, Pakistan, and Somalia:11,12,13,14,15

Tropis Field Use for fIPV
- ideal for mass vaccination campaigns
- Improved vaccine compliance
- Easy to roll out and train vaccinators
Gambia
fiPV assessment
Average doeses per vial (50 does vial)
- Needle and Syringe
50 doses per vial - Tropis
63 doses per vial
Cuba
fIPV Camaign Launch
-
- 130 Vaccinators Trained
- 100% Satisfaction
- 98% Likely to recommend
Pakistan
fIPV large scale campaign use
Campaign use launched 2019, expanded in 2020:
-
-
- Cascade Training
- 2019: 500,000 children vaccinated in 5 days
- 2020-2011:+2M children vaccinated
-
Somalia
fIPV large scale campaign use
Campaign use launched 2021:
-
- Cascae Training in Mogadishu – 110k children
- 100% parents more likely to bring child in NF
- 100% vaccinators think NF could increase compliance
- In Gambia, significant dose savings were found with the Tropis ID delivering 25% more doses out of a vial or 63 doses per vial compared to N/S which only had 50 doses per vial.
- In a study in Cuba that compared different devices, ID delivery with Tropis had higher immunogenicity vs. the Mantoux method with N/S and consistently overcame barriers to ID administration as it is easy-to-use and provides consistent delivery. Vaccinators reported 100% satisfaction and 98% were likely to recommend the NFIS.16
- In Pakistan, several campaigns sponsored by the World Health Organization from 2019-2022, as part of its polio eradication efforts, have targeted over 2.7 million children. Both vaccinators (97.6%) and caregivers (99.6%) expressed a strong preference for the Tropis System, citing ease and speed of use, appearance, and children’s response to the vaccination (less crying/ discomfort). Among caregivers, 94.7% said they would be more likely to bring their child for vaccination in a future campaign that used jet injectors. Additionally, mean vaccine coverage improved over previous IPV campaigns with an increase of 18.4%.
- In late 2021, the WHO, UNICEF and local Somalia health authorities launched a campaign to immunize children who missed their polio shots during routine immunization. The PharmaJet Tropis NFIS was exclusively used to deliver the polio vaccine, and was selected based on its proven cost savings, ease of training, and improved coverage benefits. Of the caregivers responding to a survey, 100% reported that they would be more likely to bring their child for vaccination in a future campaign that used needle-free injectors, citing the main reason as the child’s positive response to needle-free vaccination. Of the healthcare workers surveyed, 100% said that using needle-free injectors could increase vaccination rates.16
Preparing for the next opportunity
Episodes of high vaccine demand occur frequently around the world, particularly during pandemics/epidemics. The challenge of an acute antigen shortage for novel vaccines to come is highlighted by the recent COVID-19 pandemic and the monkeypox crisis. Fortunately, each year, more and more studies highlight the potential of reduced-dose immunization via the ID route. The PharmaJet Tropis NFIS addresses the shortcomings of traditional intradermal delivery methods using the Mantoux technique and is the preferred method of administration by vaccinators. Additionally, the PharmaJet Tropis ID NFIS has been scaled to nearly 8 million immunizations and has been commercialized for both polio as well as Covid vaccine delivery.
1 Banco, E et al, Biden administration declares monkeypox a public health emergency, Politico, August 4, 2022
2 Monkeypox, COVID-19 & Other Global Health Issues Virtual Press conference transcript – 27 July 2022, World Health Organization
3 Monkeypox Update: FDA Authorizes Emergency Use of JYNNEOS Vaccine to Increase Vaccine Supply, FDA News Release, August 9, 2022
4 EMA’s Emergency Task Force advises on intradermal use of Imvanex / Jynneos against monkeypox | European Medicines Agency (europa.eu)
5 Weniger, B et al, Workshop report: Nucleic acid delivery devices for HIV vaccines: Workshop proceedings, National Institute of Allergy and Infectious Diseases, Bethesda, Maryland, USA, May 21, 2015, Vaccine, Volume 36, Issue 4, 25 January 2018, pages 427-437
6 Mashunye, T et al, Fractional dose compared with standard dose inactivated poliovirus vaccine in children: a systematic review and meta-analysis, The Lancet Infectious Diseases, Vol.21, Issue 8, August 2021, pages 1161-1174
7 Schnyder, J et al, Fractional doses of intradermal compared to intramuscular and subcutaneous vaccination- A systematic review and meta-analysis, Travel Medicine and Infectious Disease, Vol 37, Sept-Oct 2020, 101868
8 Zia, S, FDA to stretch vaccine for monkeypox via intradermal injection, The Scientist, Aug. 12, 2022
9 Branswell, H and Gaffney, T, U.S. plan to stretch monkeypox vaccine supply already hitting hurdles, STAT Aug. 23, 2022
10 Daly, C. et al, Needle-free injectors for mass administration of fractional dose inactivated poliovirus vaccine in Karachi, Pakistan: A survey of caregiver and vaccinator acceptability, Vaccine, 18 February 2020, pages 1893-1898
11 Intradermal administration of fractional doses of the inactivated poliovirus vaccine in a campaign: a pragmatic, open-label, non-inferiority trial in The Gambia
12 Needle-free injectors for mass administration of fractional dose inactivated poliovirus vaccine in Karachi, Pakistan: A survey of caregiver and vaccinator acceptability – ScienceDirect
13 An experience of mass administration of fractional dose inactivated polio vaccine through intradermal needle-free injectors in Karachi, Sindh, Pakistan – PubMed (nih.gov)
14 Intradermal administration of fractional doses of the inactivated poliovirus vaccine in a campaign: a pragmatic, open-label, non-inferiority trial in The Gambia – PMC (nih.gov)
15 Feasibility of conducting intradermal vaccination campaign with inactivated poliovirus vaccine using Tropis intradermal needle free injection system, Karachi, Pakistan – PMC (nih.gov)
16 Data on file

